| [1] |
ZHANG Z M, YE J Y, LONG H F, HONG Y, LU P L. Global proteomic and phosphoproteomic analysis of the premature maize anther[J]. Journal of Biotechnology, 2016, 32(7): 937-955.
|
| [2] |
SUN Z M, CHEN Q, LI M H, MA W N, ZHAO X Y, HUANG Z. Chronic phosphoproteomic in temporal lobe epilepsy mouse models induced by kainic acid[J]. Journal of Peking University, 2019, 51(2): 197-205.
|
| [3] |
蔡冲,吕均良,陈昆松. 蛋白激酶的研究(综述)[J]. 亚热带植物科学,2002(1):63-67.
CAI Chong, LV Junliang, CHEN Kunsong. A review of study in protein kinase[J]. Subtropical Plant Science, 2002(1): 63-67(in Chinese).
|
| [4] |
张宝会,王晨桐,郭淼,肖华. 基于亲和色谱的肺癌细胞磷酸化蛋白质组研究及其应用[J]. 色谱,2021,39(1):77-86.
ZHANG Baohui, WANG Chentong, GUO Miao, XIAO Hua. Affinity chromatography based phosphoproteome research on lung cancer cells and its application[J]. Chinese Journal of Chromatography, 2021, 39(1): 77-86(in Chinese).
|
| [5] |
LUO F T, JIANG N, WANG H, SHAO X F, CHEN R B, BAI R X, WANG Y. Phosphoproteomic analysis of peripheral blood mononuclear cells of ankylosing spondylitis patients[J]. Journal of Cellular and Molecular Immunology, 2020, 36(5): 444-450.
|
| [6] |
HEAZLEWOOD J L, DUREK P, HUMMEL J, SELBIG J, WECKWERTH W, WALTHER D, SCHULZE W X. PhosPhAt: a database of phosphorylation sites in Arabidopsis thaliana and a plant-specific phosphorylation site predictor[J]. Nucleic Acids Research, 2008, 36: D1015-D1021.
|
| [7] |
NAKAGAMI H, SUGIYAMA N, MOCHIDA K, DAUDI A, YOSHIDA Y, TOYODA T, TOMITA M, ISHIHAMA Y, SHIRASU K. Large-scale comparative phosphoproteomics identifies conserved phosphorylation sites in plants[J]. Plant Physiology, 2010, 153(3): 1161-1174.
|
| [8] |
甄艳,李春映,施季森. 磷酸化蛋白质组定量研究策略[J]. 分子植物育种,2014,12(3):577-583.
ZHEN Yan, LI Chunying, SHI Jisen. Progress in quantitative analysis strategies of phosphoproteomic[J]. Molecular Plant Breeding, 2014, 12(3): 577-583(in Chinese).
|
| [9] |
JI C J, LI L. Quantitative proteome analysis using differential stable isotopic labeling and microbore LC-MALDI MS and MS/MS[J]. Journal of Proteome Research, 2005, 4(3): 734-742.
|
| [10] |
AMORESANO A, MARINO G, CIRULLI C, QUEMENEUR E. Mapping phosphorylation sites: a new strategy based on the use of isotopically labelled DTT and mass spectrometry[J]. European Journal of Mass Spectrometry, 2004, 10(1): 401-412.
|
| [11] |
GOSHE M B, CONRADS T P, PANISKO E A, ANGELL N H, VEENSTRA T D, SMITH R D. Phosphoprotein isotope-coded affinity tag approach for isolating and quantitating phosphopeptides in proteome-wide analyses[J]. Analytical Chemistry, 2001, 73(11): 2578-2586.
|
| [12] |
JI C J, GUO N, LI L. Differential dimethyl labeling of N-termini of peptides after guanidination for proteome analysis[J]. Journal of Proteome Research, 2005, 4(6): 2099-2108.
|
| [13] |
JIN M, BATEUP H, PADOVAN J C, GREENGARD P, NAIRN A C, CHAIT B T. Quantitative analysis of protein phosphorylation in mouse brain by hypothesis-driven multistage mass spectrometry[J]. Analytical Chemistry, 2005, 77(24): 7845-7851.
|
| [14] |
BARRIOS-LLERENAl M E, LE B T. Quantitative phosphoproteomic using titanium dioxide micro-columns and label-free quantitation[J]. Methods in Molecular Biology, 2019, 1977: 35-42.
|
| [15] |
ZHAI G J, WU X Y, LUO Q, WU K, ZHAO Y, LIU J A, XIONG S X, FENG Y Q, YANG L P, WANG F Y. Evaluation of serum phosphopeptides as potential cancer biomarkers by mass spectrometric absolute quantification[J]. Talanta, 2014, 125: 411-417.
|
| [16] |
SMITH J, FIGEYS D. Recent developments in mass spectrometry-based quantitative phosphoproteomics[J]. Biochemistry and Cell Biology, 2008, 86(2): 137-148.
|
| [17] |
LI Z, ADAMS R M, CHOUREY K, HURST G B, HETTICH R L, PAN C L. Systematic comparison of label-free, metabolic labeling, and isobaric chemical labeling for quantitative proteomics on LTQ Orbitrap velos[J]. Journal of Proteome Research, 2012, 11(3): 1582-1590.
|
| [18] |
SHIIO Y, AEBERSOLD R. Quantitative proteome analysis using isotope-coded affinity tags and mass spectrometry[J]. Nature Protocols, 2006, 1(1): 139-145.
|
| [19] |
朱金蕾,张锴,何锡文,张玉奎. 基于质谱技术蛋白质定量方法的研究进展[J]. 分析化学,2010,38(3):434-441.
ZHU Jinlei, ZHANG Kai, HE Xiwen, ZHANG Yukui. New development of quantitative mass spectrometrybased proteomics[J]. Analytical Chemistry, 2010, 38(3): 434-441(in Chinese).
|
| [20] |
王子健,杨静波,李光谱,孙宁宁,孙万春,彭其胜,刘宁. 低浓度甲醛对多肽和蛋白化学修饰的质谱研究[J]. 分析化学,2016,44(8):1193-1199.
WANG Zijian, YANG Jingbo, LI Guangpu, SUN Ningning, SUN Wanchun, PENG Qisheng, LIU Ning. Chemical modifications of peptides and proteins with low concentration formaldehyde studied by mass spectrometry[J]. Analytical Chemistry, 2016, 44(8): 1193-1199(in Chinese).
|
| [21] |
马首智,孙玉琳,赵晓航,徐平. 高精度相对和绝对定量的等量异位标签在定量蛋白质组学研究中的新进展[J]. 生物工程学报,2014,30(7):1073-1082.
MA Shouzhi, SUN Yulin, ZHAO Xiaohang, XU Ping. Recent advance in high accuracy iTRAQ for quantitative proteomics[J]. Chinese Journal of Biotechnology, 2014, 30(7): 1073-1082(in Chinese).
|
| [22] |
KOSAKO H, NAGANO K. Quantitative phosphoproteomics strategies for understanding protein kinase-mediated signal transduction pathways[J]. Expert Review of Proteomics, 2011, 8(1): 81-94.
|
| [23] |
ZHANG Q W, FENG X J, LI H H, LIU B F, LIN Y W, LIU X. Methylamidation for isomeric profiling of sialylated glycans by nanoLC-MS[J]. Analytical Chemistry, 2014, 86(15): 7913-7919.
|
| [24] |
LIU X, QIU H Y, LEE R K, CHEN W X, LI J J. Methylamidation for sialoglycomics by MALDI-MS: a facile derivatization strategy for both α2,3- and α2,6-linked sialic acids[J]. Analytical Chemistry, 2010, 82(19): 8300-8306.
|
| [25] |
衣泰龙,田苗苗,杨晓明,徐平. 磷酸化蛋白质组学的新进展及其在肝脏生理和病理机制中的应用[J]. 生物工程学报,2014,30(7):1004-1017.
YI Tailong, TIAN Miaomiao, YANG Xiaoming, XU Ping. Progress and application of phosphoproteomics in the proteomics of liver pathological and physiological state[J]. Chinese Journal of Biotechnology, 2014, 30(7): 1004-1017(in Chinese).
|
| [26] |
林威. 二氧化钛富集磷酸肽方法的优化及在小鼠肝脏磷酸化蛋白质组学研究中的应用[D]. 北京:北京工业大学,2012.
|
| [27] |
程功,王志刚,刘彦琳,张吉林,孙德慧,倪嘉缵. 基于纳米结构材料的磷酸化蛋白/多肽富集和分析[J]. 化学进展,2013,25(4):620-632.
CHENG Gong, WANG Zhigang, LIU Yanlin, ZHANG Jilin, SUN Dehui, NI Jiazuan. Phosphoprotein/phosphopeptide enrichment and analysis based on nanostructured materials[J]. Progress in Chemistry, 2013, 25(4): 620-632(in Chinese).
|
 本站查看
本站查看



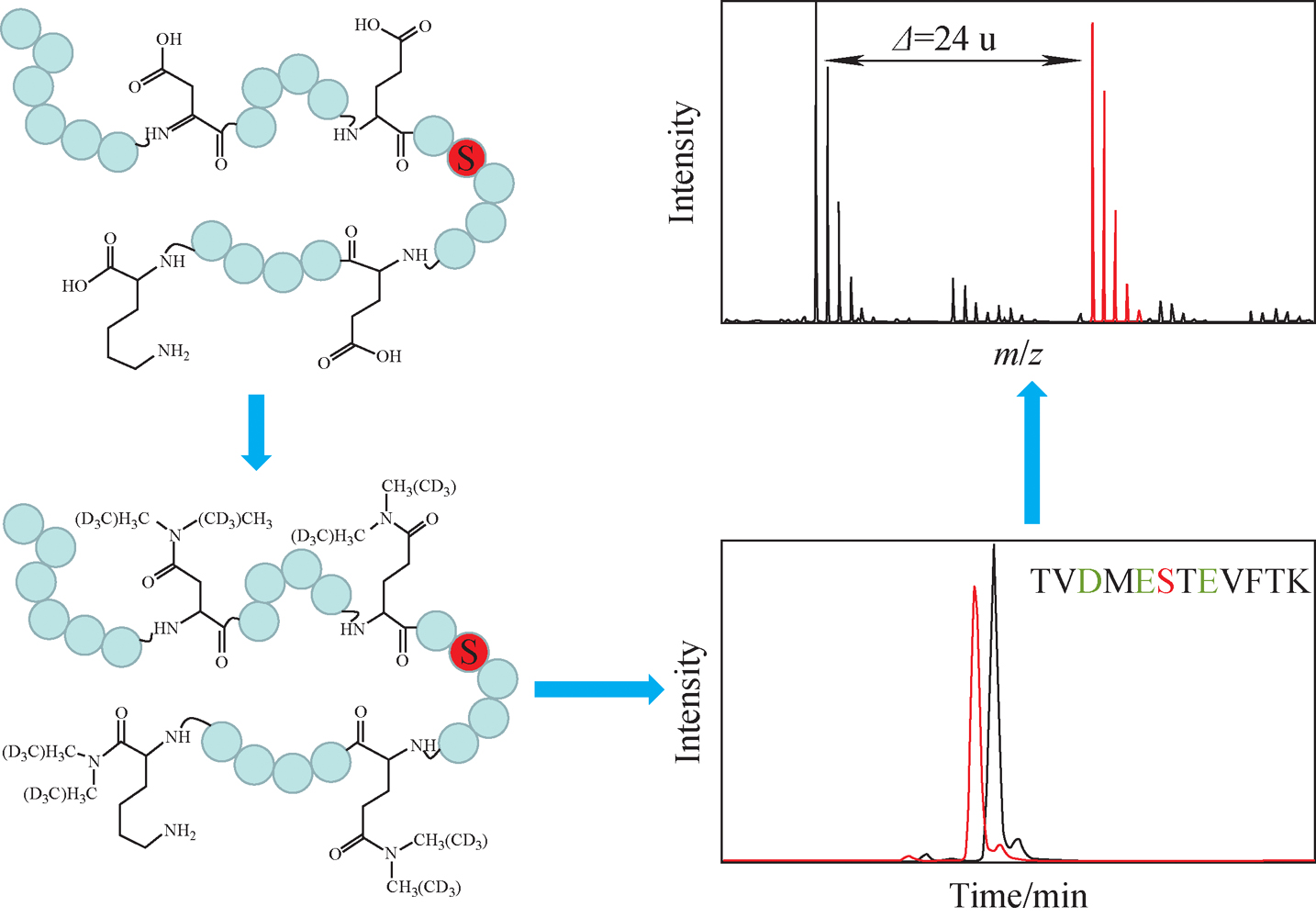
 下载:
下载:
